 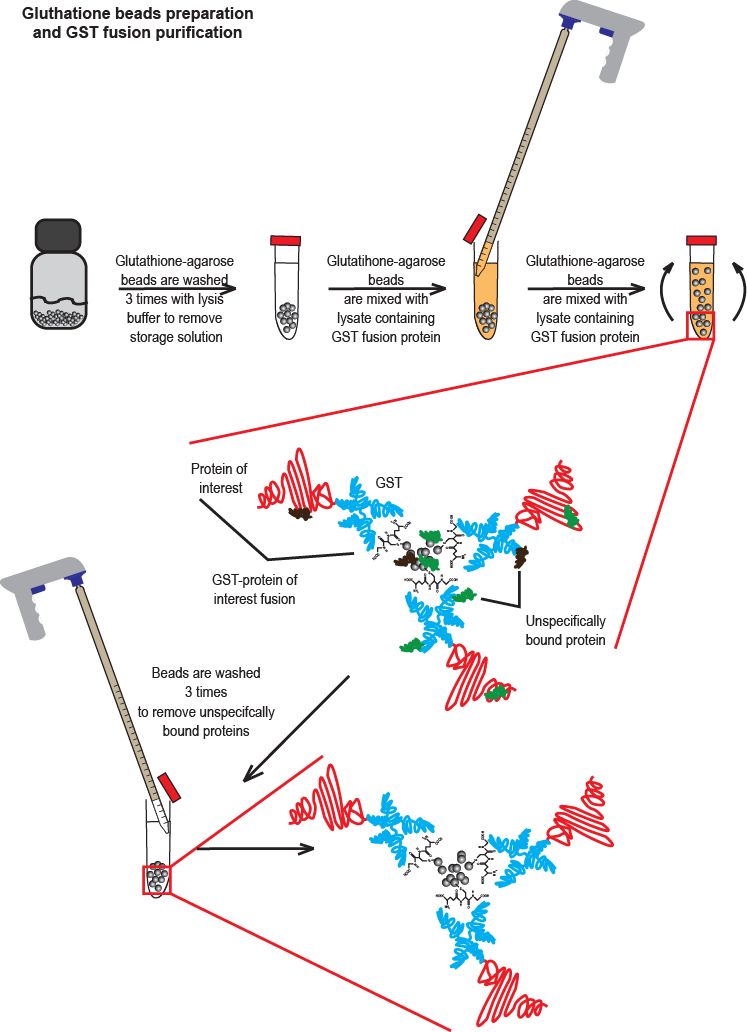
Transfer any other protein samples to clean pre-labeled microcentrifuge tubes and bring to optimal concentration with UltraPure Sterile Water.Add reducing agent (β-ME, DTT or TCEP) as needed and mix well by pipetting. If using a lysate already in Sample Buffer, thaw lysate and transfer 25 µL of lysate to a clean pre-labeled microcentrifuge tube.Use 4-20% gels to separate proteins 10 to 200 kDa in size.Use 4-8% gels to separate proteins 100 to 500 kDa in size.Determine the best gel to use according to the molecular weight (MW) of the protein of interest:.Procedure for SDS-PAGE Sample Preparation and Separation Note: Extracts prepared in this manner may be stored for months at -70☌ without appreciable degradation of target when avoiding repeating freeze/thaw cycles. Snap-freeze the supernatant in labeled, chilled tubes store at -70☌ until needed for electrophoresis, transfer, and detection steps.Note: The protein concentration of each sample should be determined so that the amounts of proteins from the different samples can be compared. When resuspended in 2X SDS-PAGE Sample Buffer, determining protein concentration is difficult due to the presence of interfering compounds with most colorimetric protein assays. If 1X RIPA Lysis Buffer was used, take a 10-µL aliquot of each extract and determine protein concentration.Transfer the resulting supernatants (whole cell lysates), which contain the protein to be examined, to fresh, precooled microcentrifuge tubes.Clarify by high-speed centrifugation for 10–15 minutes at ∼15,000 × g, 4☌.Disrupt cells and shear DNA by sonication using two 7-second, 50W pulses with a 20-second interval per ∼0.5-mL sample on ice. 
Dislodge cells using a cell scraper and transfer to a tube.Note: Alternatively, non-stimulated cells can be trypsinized and washed with ice-cold PBS before lysis. Add 0.5 mL of ice-cold 1X RIPA Lysis Buffer or 2X SDS-PAGE Sample Buffer per 1 x 10 7 cells (approximately 0.5 mL for a sub-confluent 100 mm plate or 75 cm 2 flask, 0.7 mL for a 150 cm 2 flask).Remove growth medium, gently rinse cells with ice-cold 1X PBS.Grow cells to optimal confluency in appropriate growth medium.Mix the two components in a 1:1 ratio just before use. Make Blocking Buffer by adding Tween-20 to a final concentration of 0.1%.ġ0X TTBS pH 7.5 (1.0 M Tris HCl 1.5 M Sodium Chloride 0.1% (v/v) Tween-20)ġ0X TBS pH 7.5 (1.0 M Tris HCl 1.5 M Sodium Chloride)Īnti-Rabbit IgG (H&L) (Goat) Antibody Peroxidase ConjugatedĪnti-Mouse IgG (H&L) (Goat) Antibody Peroxidase ConjugatedĪnti-Goat IgG (H&L) (Rabbit) Antibody Peroxidase ConjugatedĬhemiluminescent FemtoMax™ Super Sensitive HRP Substrate for Microwell and/or Membrane Opal Prestained Protein Standard 10-245kDa or 10-180kDa Keep on ice.ġ0X SDS-PAGE Running Gel Buffer or 10X Tris-Glycine Make 1X solution by diluting with UltraPure Sterile Water. Keep on ice.Īppropriate transfer cell and power supplyĢX SDS-PAGE Sample Buffer without DTT or β-ME Endogenous, stimulated and/or transgenic cell lysates.Īdd 1X protease and phosphatase inhibitors just before use. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
